The WATCHMAN FLX™ LAAC Device met all trial endpoints as a first-line option vs. NOACs, including a superior net clinical benefit*, 1.1% annualized ischemic stroke rate and 45% bleeding risk reduction.
These findings reinforce Boston Scientific’s LAAC leadership and strengthen WATCHMAN’s unmatched clinical evidence supporting LAAC as an option for lifelong stroke risk reduction without the bleeding risk of long-term oral anticoagulation.
*Net clinical benefit endpoint includes a composite of cardiovascular death, stroke, systemic embolism, and non-procedural ISTH major and modified clinically-relevant non-major bleeding
CHAMPION-AF Trial Design
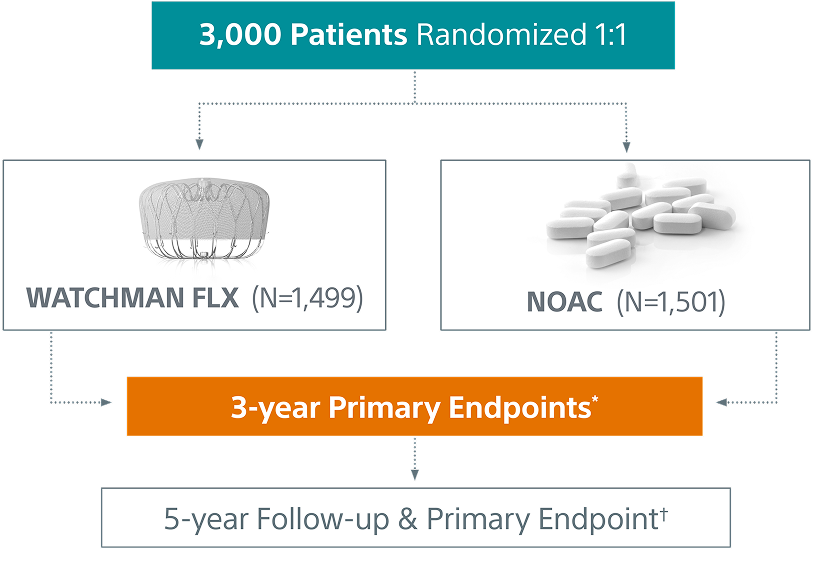
*study success is defined as meeting both 3-year primary endpoints
†5-year primary endpoint is non-inferiority for the occurrence of ischemic stroke or systemic embolism
CHAMPION-AF Trial Design
CHAMPION-AF is a prospective, randomized, multi-center, global investigation to study left atrial appendage closure with the WATCHMAN FLX™ LAAC Device as a first-line alternative to oral anticoagulation in a broad non-valvular atrial fibrillation population, including those who are at low-to-moderate risk of bleeding from the use of oral anticoagulation.
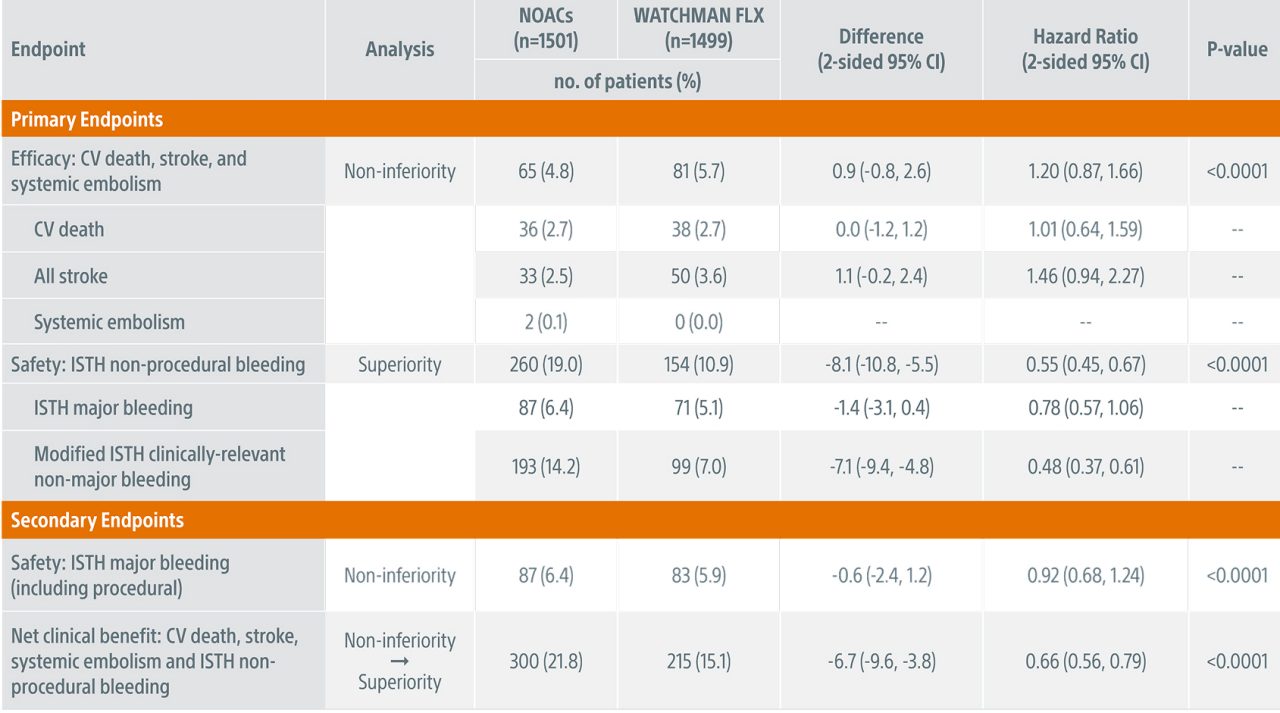
CHAMPION-AF Primary Endpoint Outcomes (3-years)
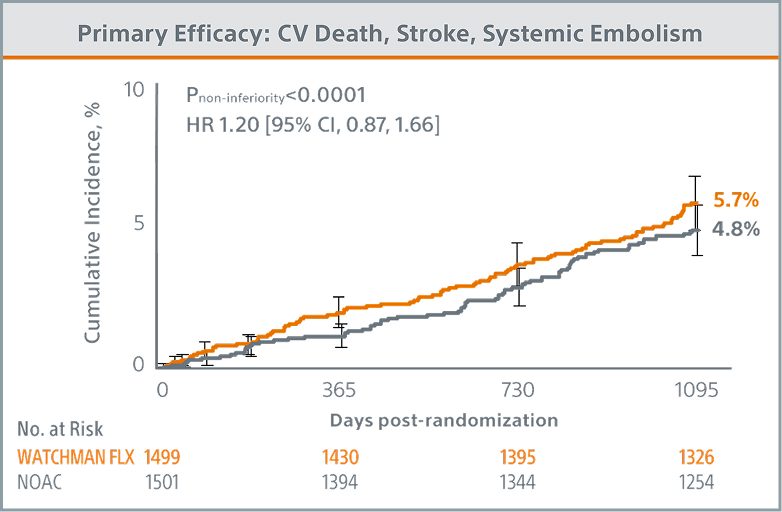
Primary Efficacy Endpoint Met
WATCHMAN FLX demonstrated statistical non-inferiority to NOACs for the occurrence of cardiovascular (CV) death (hemorrhagic and/or unexplained death), stroke (ischemic and/or hemorrhagic), and systemic embolism (5.7% vs. 4.8%; Pnon-inferiority <0.0001)
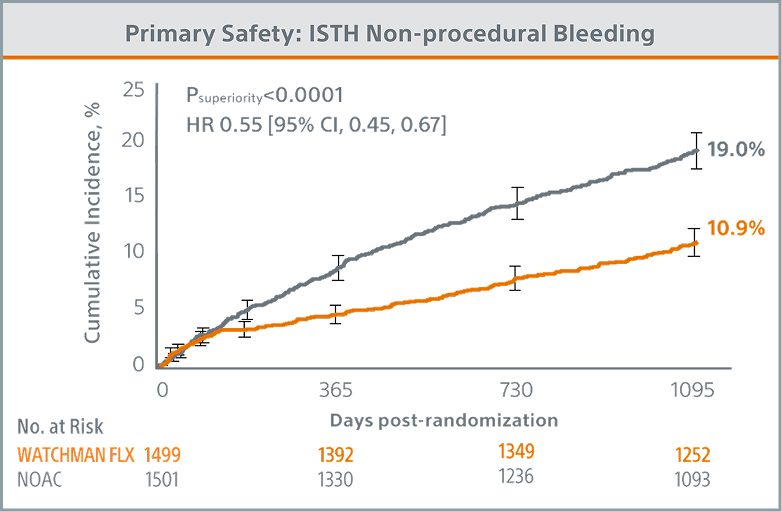
Primary Safety Endpoint Met
WATCHMAN FLX demonstrated statistical superiority to NOACs for the occurrence of ISTH non-procedural major bleeding and modified* clinically relevant non-major bleeding (10.9% vs. 19.0%; Psuperiority <0.0001)
*Modified ISTH clinically relevant non-major bleeding was defined as any sign or symptom of hemorrhage (e.g., more bleeding than would be expected for a clinical circumstance, including bleeding found by imaging alone) that does not fit the criteria for the ISTH definition of major bleeding but does meet at least one of the following criteria.
▪ Requiring medical intervention by a healthcare professional
▪ Leading to hospitalization or increased level of care (e.g., ER visit, diagnostic procedures, medication change)
Primary & Secondary Endpoints (Kaplan-Meier Estimates)
Ischemic Stroke/Systemic Embolism (SE) Rates Across Landmark Trials
Stroke rates from both the WATCHMAN FLX and NOAC arms were low. The WATCHMAN FLX device demonstrated a 1.1% annualized ischemic stroke/SE rate, aligned with ischemic stroke rates observed in prior, seminal clinical trials of NOACs, suggesting both strategies may be effective for patients seeking a stroke risk reduction therapy.
 CHAMPION-landmark-chart
CHAMPION-landmark-chart
Results from different clinical investigations are not directly comparable. Information provided for educational purposes only. CHAMPION-AF NOAC arm annualized ischemic stroke/SE rate: 0.7%
† CHA₂DS₂-VASc score used in CHAMPION-AF trial only; CHADS₂ score used in NOAC trials
ISTH Bleeding (Including Procedural Bleeding):
With the inclusion of procedural bleeding, WATCHMAN FLX demonstrated a statistically significant 34% relative reduction in ISTH major and modified clinically relevant non-major bleeding at 36 months (12.8% vs. 19.0%; P<0.0001).
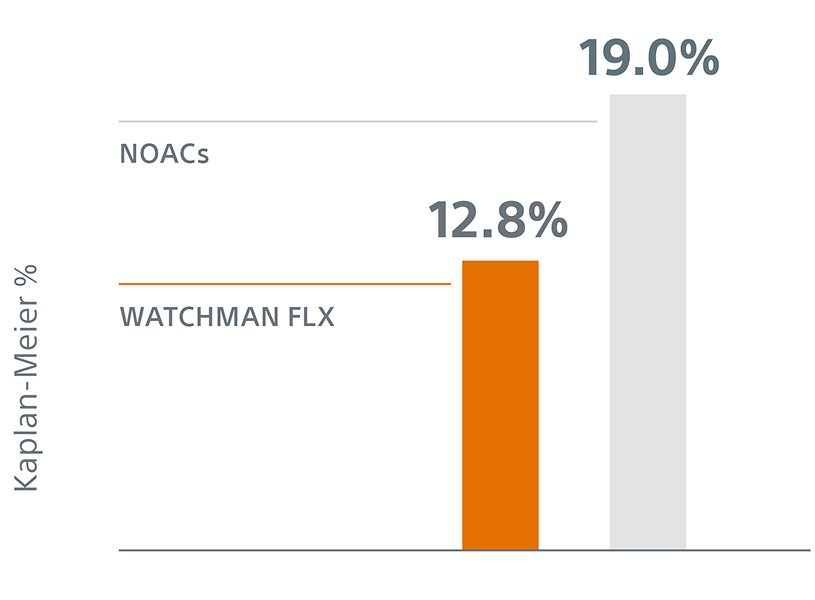
The 34% reduction (HR 0.66 [0.54, 0.80]) in ISTH bleeding (including procedural) at 36 months further reaffirms superiority of WATCHMAN FLX for bleeding risk reduction compared to NOACs.
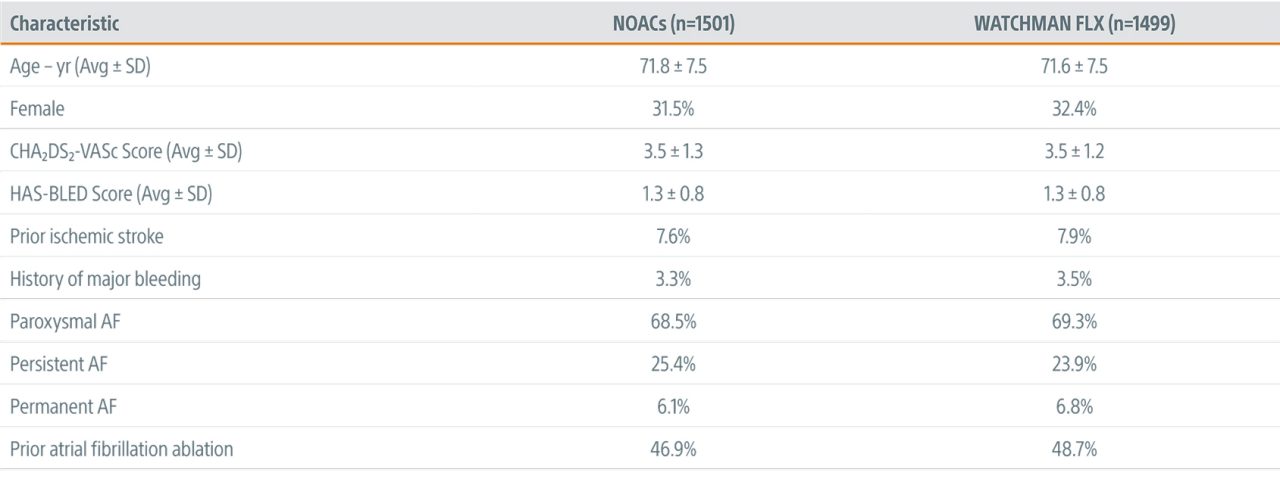
Key Baseline & Procedural Characteristics
Procedural success* across 141 implanting centers reinforces the simplicity of the WATCHMAN FLX platform on a global scale
*Device successfully deployed and released
Learn about the first randomized, head-to-head study comparing WATCHMAN to OACs after cardiac ablation
References
- SK Doshi et al. Left Atrial Appendage Closure or Anticoagulation for Atrial Fibrillation. New England Journal of Medicine, March 2026
- Patel, M. Rivaroxaban versus Warfarin in Nonvalvular Atrial Fibrillation. NEJM 2011; 365(10):883-891.
- Granger, C. Apixaban versus Warfarin in Patients with Atrial Fibrillation. NEJM 2011; 365(11):981-992
- Connolly, S. Dabigatran versus Warfarin in Patients with Atrial Fibrillation. NEJM 2009; 361(12): 1139-1151
- Giugliano, R. Edoxaban versus Warfarin in Patients with Atrial Fibrillation. NEJM 2013; 369(22): 2093-2104.
WATCHMAN FLX is an FDA approved device being studied for an expanded indication as a first line therapy vs NOAC for NVAF patients. The use of WATCHMAN or WATCHMAN FLX as a first-line therapy for stroke risk reduction in NVAF patients is considered investigational. Caution: Investigational Device. Limited by US law to investigational use only.
